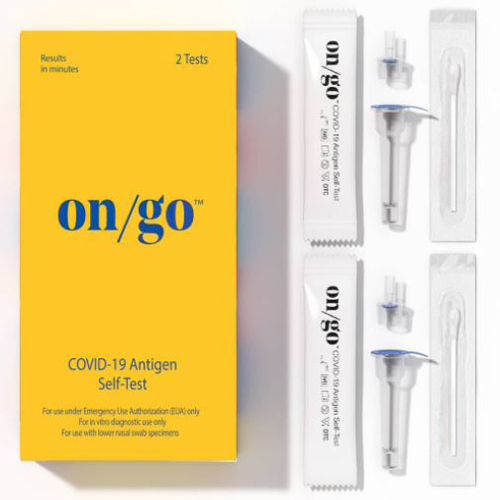
Description
- FDA Emergency Use Authorization
- Rapid results at 10 minutes
- 95% accurate (PPA: 87%, NPA: 98%)
- Detects the antigen protein of all known major COVID-19 variants, including Delta and Omicron
- Authorized for non-prescription self-use for individuals 14 years or older or adult-collected samples from individuals 2 years or older
- For individuals with or without symptoms
- Qualitative detection of SARS-CoV-2 nucleocapsid protein antigen via lower nasal swab samples
- For in vitro diagnostic use only
- 12 month expiration from manufactured date
- Made in USA